Gravity Entropy Theory
1. The Gravity-Entropy
Theory states that on a scale from molecules to stars, gravity is the dominant
attractive force, and entropy, is a repulsive force that opposes
gravity. The textbook equations for entropy are in part correct, but are
incomplete. Dr. Klein postulated that entropy is not merely a scalar
(a quantity), It is a vector (it also has direction), but the direction of the entropy
vector is opposite that of gravity.
Entropy governs phenomenon
such as temperature, pressure, and chemical concentration. Entropy is a
repulsive force, pushing matter and energy outward or apart—from areas of
greater temperature, pressure, or chemical concentration to areas of lesser
temperature, pressure, or chemical concentration toward equilibrium. Gravity,
on the other hand, pulls matter inward—in a direction opposite to that of
entropy. Gravity pulls matter from areas of lesser concentration
toward the center to areas of greater temperature, pressure, and mass
concentration.
2. Gravity Equation: G= m1m2/r2
This means that the Force of Gravity is directly proportional
to the mass of the first body times the mass of the secong body, and is
inversely proportional to the square of the distance between the masses. Thus, the greater the distance between the
masses, the much weaker the gravitational force.
3. Entropy Equation:
In chemistry, entropy is
represented by the capital letter S, and it is a thermodynamic function that
describes the randomness and disorder of molecules based on the number of
different arrangements available to them in a given system or reaction.
The amount
of entropy is also a measure of the molecular disorder, or
randomness, of a system
The American
Heritage Dictionary gives as the second definition of entropy,
"a measure of disorder or randomness in a closed system."
The change in entropy, ∆S, is
given by : .
∆S = ∆Q
, or
T
Change in entropy = change in the heat of the system
Temperature
Where
∆S = change in entropy (joules/Kelvin or calories/Kelvin)
∆Q = change in
the heat of the system (joules or calories)
T = temperature (Kelvin)
4. The textbook
definitions and equations for entropy are correct; they are just
incomplete. They define entropy as a
scalar (a number), which for a system always appears to be increasing. Entropy has not only a numeric value, it also
has a direction.
Thus, according to Dr. Klein’s theory, entropy is a vector.
Entropy is a force; it has a magnitude and a direction. The direction is outward, opposing that of
gravity, which is inward.
The entropy vector goes from greater to lesser toward
equilibrium.
Gravity is an attractive force, pulling matter inward toward
the center of mass. Entropy is a
repulsive force, pushing outward toward equilibrium. Entropy is an equilibrium force, pushing
matter and energy outward toward equilibrium.
5. Gravity and entropy are opposing forces. There is a balance in nature. For every attractive force, there is also a
repulsive force that tends to balance it.
The repulsive force is not necessarily exactly equal to the attractive
force. The balance between gravity and
entropy is not a static balance; it is a dynamic balance—matter and energy are
constantly moving and changing.
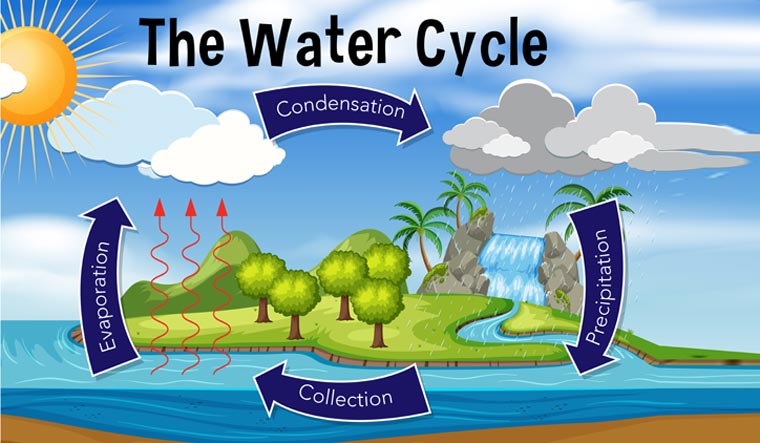
6. An example of the
dynamic balance between gravity and entropy is the water
cycle. Entropy causes water to evaporate, going from greater vapor
pressure to lesser vapor pressure in the atmosphere. Upon
condensation, gravity pulls the rain toward the earth’s center, causing the
rain to fall back to the earth.
7. Other examples.
Letting air out of a tire, such a by a puncture. Pressure goes from greater to lesser toward
equilibrium. Gravity pulls air in the
atmosphere downward toward the earth’s center, while entropy pushes it outward
toward outer space where the pressure is less.
Another example is when chemicals are mixed in a mixture,
the chemical concentration goes from greater to lesser areas of concentration
toward equilibrium.
8. Background microwave radiation. The primary source of the microwave radiation
is the stars. Entropy is an equilibrium
force that causes the causes the microwave radiation to be roughly evenly
distributed in space.
Thus, the cosmic background
radiation is not due to a fictitious Big Bang (see Cumulative Field
Theory). The true source of the cosmic background radiation is the
stars and dark matter in space (planets, moons, asteroids, etc.) that radiate
energy into space. Then, entropy causes the radiation to go from
areas of greater temperature or greater concentration to areas of lesser
temperature or lesser concentration toward equilibrium. This results
in a roughly even temperature distribution in space. |